 
This was found true even when the masses were very different. He was trying to understand why in chemical reactions involving gases the observation that equal volumes of different gases had the same number of moles. 
Avogadro was an Italian Physicist who first described the Avogadro constant as a hypothesis in 1811. If I assume that we are mostly water on the average because our average density is approximately that of water then I can use the above information on water to get the answer. A pound is 454 grams so a 10 pound human head is 4,540 grams. Armed with all this we can estimate the number of atoms in your head. 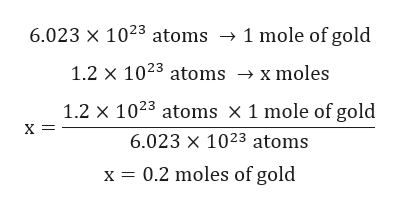
This observation will lead you to conclude that our density is very close to the density of water. When you go swimming you probably noticed that almost everybody floats with just part of their head out of water. A typical human head weighs about 10 - 12 pounds. Now we know enough to answer your question. The number of atoms is 3 times larger because each molecule has three atoms, so there are 1.0038*10 26 atoms in a liter of water. Hydrogen has an atomic weight of 1 and oxygen has an atomic weight of 16. Water is made up of 2 hydrogen atoms and one oxygen atom. Water is easy because each cubic centimeter has 1 gram of mass. A liter of water is 1000 cubic centimeters. If you know how heavy something is or what its volume and density are you can easily do this. 
In the above formula density times volume is just the mass. N is a constant called Avogadro's number and its equal to 6.022*10 23 atoms/ mole. # of atoms = N * (density) * volume / (Molecular Weight). The number of atoms of ANY substance in a volume is: This is really just an estimate, but it's going to be a good one. This is the volume of the substance, which can be converted into any volumetric units.We can calculate the number of atoms in your head if we know the density and a constant called Avogadro's number. Density of SubstanceĮnter the known density of the material being measured. The formula used by this calculator to determine volume from mass and density is:Įnter the measured mass of the object and select the appropriate mass measuring units. This calculator will determine the volume of a quantity of substance from the measured mass and known density and display a conversion scale for variations in each parameter.įor example this tool can be used to calculate the amount of storage volume required for a given quantity of substance mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
